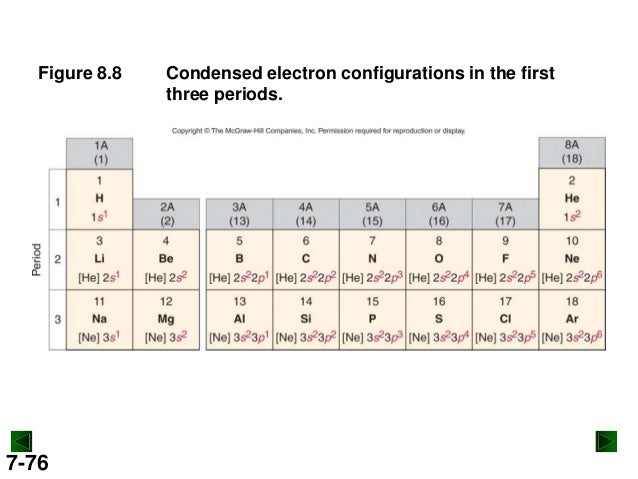
By forcing electrons to occupy different orbitals, electron-electron repulsion is minimized. Hund's rule of maximum multiplicity states that the greater total spin states results in a more stable configuration. Experimentally Cr is found to be 3d 5 4s 1.Īccording to Pauli's Exclusion principle no two electrons of the same atom can occupy the same set of quantum numbers (n, l, m l, m s), therefore electrons of m s differ by having opposite spins. The expected electron configuration of Cr is 3d 5 4s 1. Chromium and copper have ground states that are exceptions to the Aufbau. It is important to note that Aufbau is not accurate every time. Generally, the electrons are filled up in this order: 1s 1, 2s 2, 2p 6, 3s 2, 3p 6, 4s 2, 3d 10, etc. The Aufbau principle (Aufbau meaning "build up" in German) postulates the electrons "build up" by placing two electrons in each orbital filling up orbitals from lowest energy to highest. There are also some rules regarding electron configurations that you must be familiar with. Thus the quantum state is described by the set (n, l, m l, m s).  
Spin quantum number, m s, there can be two different spins can fit per orbital designated as +1/2 or -1/2.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
